Your Questions, Answered
We’ve assembled the most frequently asked questions about Eversense into one place. Got a question that you can’t find the answer to? Contact us.
There are three steps to ensure proper positioning:
- When using the adhesive patch to secure the smart transmitter, make sure the power button and the LED are lined up in parallel with the sensor.
- Use the PLACEMENT GUIDE screen on the app to confirm connection between the sensor and the transmitter.
- Tap Placement Guide.
- Position the smart transmitter over the sensor so that a Good or Excellent connection is confirmed. Allow time for the Placement Guide to refresh – about 0.5 seconds.
For more details on the signal strength of the transmitter and sensor, tap “Show More Details”.
This feature is to be used for troubleshooting and not intended as first course of action for the user.
The smart transmitter is incompatible with magnetic resonance imaging (MRI) procedures. The smart transmitter is MR Unsafe and MUST BE REMOVED before undergoing an MRI (magnetic resonance imaging) procedure.
The system is contraindicated in people for whom dexamethasone or dexamethasone acetate may be contraindicated.
Mannitol or sorbitol, when administered intravenously, or as a component of an irrigation solution or peritoneal dialysis solution, may increase blood mannitol or sorbitol concentrations and cause falsely elevated readings of your sensor glucose results. Sorbitol is used in some artificial sweeteners, and concentration levels from typical dietary intake do not impact sensor glucose results
Manual BG entries cannot be edited. Meals, Insulin, Health, and Exercise icons may be edited by going to the Event log and tapping on the entry.
An event icon can be modified or hidden after it was previously saved. From the Event Log, click on the event icon you would like to modify. Once the changes are made, tap Save.
To remove an event icon, select the event icon you would like to modify, scroll down to the bottom of the page and select “Hide Event”. This will remove the event icon from the home page.
Manual BG Entry:
- Can be hidden
- Cannot be edited
- Cannot be deleted
Events:
- Can be hidden
- Can be edited
- Cannot be deleted
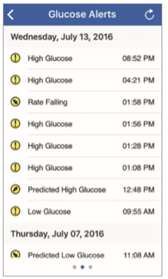
To view the Eversense® CGM user’s Glucose Alert History, swipe your screen to the left from the trend graph.
The Alert History screen will list the last 20 glucose-related alerts that the user received in their Eversense® CGM App.
These alerts monitor events such as the rising or falling of glucose rates as well as low or high, and predicted low or high, glucose values (hypoglycemia or hyperglycemia).
Note: You will only receive predicted high and low glucose alerts if the Eversense® CGM user has predictive alerts enabled.
While the Eversense® E3 Sensor is intended to be removed at end of sensor life, it has undergone biocompatibility testing and meets the requirements of a permanent implant, and therefore it should not cause a biocompatibility concern if left in a patient. However, the sensor will not provide data beyond 180 days and you will need to make arrangements to have it removed.
A compatible smart device is required in order to use the Eversense E3 CGM. Please see our Compatibility page for a list of compatible devices.
Still have questions?
The Eversense team is dedicated and ready to provide the answers and support you need. Simply click the link below.
The Eversense® E3 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to 180 days in persons with diabetes age 18 and older. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are still required for calibration primarily one time a day after day 21, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense E3 CGM System is a prescription device; patients should talk to their health care provider to learn more.
The Eversense® 365 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to one year in people (18 years and older) with diabetes. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are required for calibration one time a week after day 13, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense 365 CGM System is a prescription device; patients should talk to their health care provider to learn more.
For important safety information, see bit.ly/eversensesafety
Eversense, Eversense E3 Continuous Glucose Monitoring, Eversense 365 Continuous Glucose Monitoring, and the Eversense logo are trademarks of Senseonics, Incorporated. All other trademarks are properties of their respective owners and are used solely for informative purposes. No relationship or endorsement should be inferred or implied.
Apple Watch® is a product of Apple, Inc., and may be separately purchased from an authorized Apple retailer. Apple Watch is not included with the Eversense CGM System. Android is a trademark of Google LLC.
MKT-001692 Rev 1